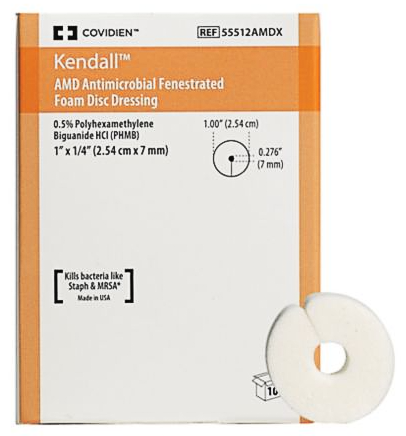
Absorbent, non-adherent, semi-occlusive, polyurethane foam disc impregnated with broad-spectrum antimicrobial (polyhexanide, 0.5%). Double-sided with centre hole and radial slit. Antimicrobial barrier is effective for up to 7 days.
Designed to provide protection and cushioning at surgically induced body exit sites such as those associated with IV catheters, central venous lines, arterial catheters, dialysis catheters, peripherally inserted coronary catheters, mid-line catheters, tracheostomy, G-tube, J-tube, Penrose drain, chest tube, nephrostomy tube, sump drain, orthopaedic pins and epidural catheters. Provides local exudate management and the Polyhexanide helps prevent bacterial colonisation of the foam dressing.
Can be used in conjunction with prescribed therapies for the treatment of infection. Not intended as a primary treatment for infection. Do not use as a primary treatment for third-degree burns. Do not use on patients with known sensitivity to polyhexanide.


A sterile dressing containing the antimicrobial substance polyhexamethylene biguanide (Polyhexanide, polyhexanide), which kills bacteria and inhibits its growth. Polyhexanide released in the presence of wound exudate is an effective antimicrobial agent against a broad spectrum of microorganisms. Manages wound exudate and microbial contamination, reducing wound pain during healing. The perforated wound contact layer contains a gentle silicone adhesive that provides secure, non-irritating adhesion and supports non-traumatic removal during dressing changes. The low-friction, waterproof polyurethane film provides a bacterial barrier to the wound.


